Joint pain in children
Septic arthritis, reactive arthritis and psoriatic arthritis
Juvenile Arthritis
 Juvenile Arthritis (JA) describes several inflammatory, autoimmune and rheumatic diseases that can develop in young individuals.
Juvenile Arthritis (JA) describes several inflammatory, autoimmune and rheumatic diseases that can develop in young individuals.
This article will discuss only septic arthritis, reactive arthritis and psoriatic arthritis.
‘Arthropathy’ is a term used to describe a dysfunctional joint- generally (but not exclusively) whereby an inflammatory process is involved1. ‘Uni’ arthropathy is used to describe a condition affecting one joint, whilst ‘poly’ arthropathy is used to describe a condition affecting two or more joints2. This differentiation can aid the practitioner to formulate a diagnosis – some conditions are more likely to present as uni-arthropathic (single joint involvement) while others more likely to be poly-arthropathic (effect multiple joints)2.
Septic Arthritis
Septic Arthritis results from microbial agents/microorganisms infiltrating the joint and causing infection3. Hemategenous spread (transfer of infection through the blood stream) into the lining of the joint (synovium) is the most common route of microorganism infiltration4. Less commonly the microorganisms can enter via direct penetration (trauma) or via contact with adjacent infected bone (osteomyelitis)4.
Because of their developing bony anatomy, juveniles can often have coexisting osteomyelitis and septic arthritis4. This is due to the thin bony cortex and ‘loose’ periosteum, whereby blood vessels connecting the cartilage growth centres can serve as a passage by which osteomyelitis (bone infection) can reach the joint space more freely than in adults4.
Inflammatory cells break-down and damage joint cartilage4 while increasing inflammatory transudate (fluid) leads to increased pressure within the joint space. This in turn compresses the intra-articular blood vessels and reduces blood supply to the cartilage and adjacent bone4. This pressure-necrosis can destroy joint synovium and cartilage.
Progression of infection can lead to avascular necrosis (tissue death) of the bony tissue (the femoral head) as well as ligamentous damage, joint dislocation and osteomyelitis4.
Common pathogens in septic arthritis4
Age |
Infectious Agent |
| Neonate |
|
| 2 months – 5yrs |
|
| Adolescents |
|
Presentation of septic arthritis
Severe pain around the joint which may be accompanied by swelling. In cases effecting joints of the lower limb, parents may report that the child is unwilling to weight-bear or is doing so with an exaggerated limp. In the acute setting movement of the affected joint in all directions may be painful.
Examination
- Reduced joint range of movement
- Tenderness around the joint margins
- Palpable warmth around the joint
- Swelling and redness of the joint – absence of this does not exclude septic arthritis.
It is also common for the child to place the joint in a position that is least painful – the hip tends to be flexed, abducted and externally rotated while the shoulder tends to be held in a position of adduction and internally rotation. The knee and elbow may be held in mild flexion whilst the ankle can be held in a ‘relaxed’ plantar flexed position.
A retrospective study by Kocher et al5 found that the strongest variables distinguishing septic arthritis from transient synovitis (discussed below) were:
- A history of fever
- Difficulty weight-bearing on the affected limb
- ESR >40mm/h (erythrocyte sedimentation rate) White blood cells >12000 cells/uL.5
Treatment of septic arthritis
All juveniles with expected septic arthritis should be hospitalized for empiric intravenous antibiotic therapy. Following splinted immobilization in a functional position for 2-3 days, early passive joint range of motion exercise can be commenced6. The duration of antibiotic therapy is dependent on the causative organism, concomitant systemic illness or compromised immune status as well as the duration of symptoms and the associated tissues infected.
In joints other than the hip, repeated needle aspirations may be conducted until fluid ceases to accumulate6. Should fluid continue to accumulate or complications arise following 3 days, arthrotomy (opening of the joint) and drainage should be considered6. If septic arthritis of the hip is suspected, consultation with an orthopaedic surgeon is indicated – the joint will require irrigation and drainage in order to avoid aseptic necrosis of the femoral head6. Consultation with infectious disease specialists may also be indicated in the presence of uncertain diagnosis or unusual microbiology results6.
Reactive arthritis
 Reactive arthritis (ReA) is an autoimmune response to infection7. ReA has commonly been associated with genitourinary infections, particularly Chlamydia trachomatis8, and gastrointestinal infections including Campylobacter, Shigella and Salmonella9. ReA is commonly classified as post-dysenteric or post-venereal based on whether the infectious origin was gastrointestinal or genitourinary.
Reactive arthritis (ReA) is an autoimmune response to infection7. ReA has commonly been associated with genitourinary infections, particularly Chlamydia trachomatis8, and gastrointestinal infections including Campylobacter, Shigella and Salmonella9. ReA is commonly classified as post-dysenteric or post-venereal based on whether the infectious origin was gastrointestinal or genitourinary.
Reactive arthritis may be associated with urethritis (inflammation of the urethra) and conjunctivitis (inflammation of the conjunctive of the eye). It I important to note that these are not found in all cases of ReA10.
Human leukocyte antigen (HLA-B27) is a genetically acquired antigen and is associated with ReA. This classifies it in the category of ‘seronegative spondyloarthropathies’ which also includes Ankylosing Spondylitis, Psoiatic arthritis and ‘undifferentiated spondyloarthritis’11. ReA and Ankylosing Spondylitis have been seen to display similar symptoms including sacroilitis (inflammation of the sacroiliac joint) and iritis (Inflammation of the iris of the eye).
ReA has a significant genetic component – it appears to cluster within families and predominantly affects young males11. HLA-B27 is observed in 70-80% of cases of ReA.
ReA typically follows a self-limiting course. Symptoms often resolve within 3-12 months14. Mortality, though rare, is more frequently associated with the adverse effects of treatment. Post-dysenteric cases are notably less severe than post-venereal cases. The presence of HLA-B27 as well as infections caused by Yersinia, Salmonella, Shigella, or Chlamydia is associated with a more protracted and severe illness.15. The recurrence rate is 15-50%, and is increased in HLA-B27 positive individuals. Between 15 and 30% of individuals may suffer long-term erosive arthritis, enthesitis or spondylitis.
Asymmetrical, lower limb poly-arthropathy is most often the presenting complaint in cases of reactive arthritis. Symptoms may be accompanied by fever, malaise and fatigue16. Acute myalgia (muscle aches and pains) may also be present. Lumbar back pain is reported in approximately 50% of presentations. Plantar fascioses (pain at the sole of the heel) and achilles enthesoses (pain at the rear of the heel) are also common.
Urogenital symptoms present in 90% of ReA cases. Post-dysenteric and post-venereal ReA may both present initially as ‘non-gonococcal urethritis’. Symptoms may include:
- urethral discharge
- urinary urgency (an urgent need to urinate)
- frequency (increased toileting) and
- dysuria (pain with urination)
Other symptoms of ReA include muscle and joint pain, photophobia (sensitivity to light), erythema (skin redness), increased tear production and impaired vision.
Reactive arthritis is associated with immune dysfunction as may be seen in cases of HIV. New onset reactive arthritis may be screened for HIV.
Examination
Two or more of the following must be exhibited for ReA to be diagnosed, with at least one symptom pertaining to the musculoskeletal system:
- Asymmetric poly-arthropathy (commonly of the lower limbs)
- Enthesitis (inflammation of the tendon-bone junction), dactylitis (inflammation of the fingers or toes), or heel pain
- Cervicitis (inflammation of the neck of the uterus)
- Diarrhoea within 1 month of onset of arthritis
- Conjunctivitis or iritis (inflammation and redness of the eye)
- Urethritis or genital ulcers
Treatment
There is no curative treatment for reactive arthritis. Symptom relief and supportive care are the primary goals of management. Approximately two-thirds of cases are self-limiting (do not progress to more severe or long lasting symptoms), and around one-third of cases will develop chronic symptoms.
Physiotherapy and non-steroidal anti-inflammatory dregs (NSAIDs) are indicated for joint, tendon and fasciae pain. Antibiotics may be prescribed to treat the underlying infection, and anti-rheumatic DMARDs (Disease Modifying Anti-Rheumatic Drugs) such as Methotrexate and Plaquenil (Hydroxychloroquine) may be beneficial.
 Hospitalisation is indicated when:
Hospitalisation is indicated when:
- Individuals are unable to tolerate oral medication.
- Concomitant severe systemic disease or immunocompromisation exists.
- Severe joint pain prevents ambulation.
Psoriatic Arthritis
Psoriasis is a disease whereby the immune system is ‘triggered’ to ‘target’ the skin. This causes scaly red lesions to form18. Psoriatic Arthritis (PA) is an inflammatory condition that develops in about 15% of individuals who have Psoriasis19 and can cause marked and prolonged joint pain. This condition may affect any joint in the body and symptoms vary significantly from case to case.
Untreated PA may cause persistent inflammation and lead to severe and permanent damage of the affected joint. Although most commonly effecting people between the ages of 30 to 5020, PA may develop in children. In rare cases arthritis (joint pain and inflammation) can precede dermatological symptoms21. More commonly a psoriatic rash will appear months or even years before joint symptoms are experinced21.
The symptoms of PA may flare and subside. One or multiple joints may be affected, and presentation can be symmetrical or asymmetrical21. Psoriatic arthritis my also affect spinal joints, ligament and tendon tissues21, 22. Enthesitis is one characteristic of psoriatic arthritis that may aid in differentiation from other conditions22.
Generally Psoriasis is diagnosed following blood tests and skin biopsy. Psoriatic arthritis can be diagnosed using pattern of affected joints, presence of enthesitis, skin and nail changes found in psoriasis and imaging to assess joint structure.
Presentation
The diagnosis of PA is made by clinical examination. Findings should include psoriasis (red scaly, dry plaques or rashes) and joint inflammation of the spine and/or peripheral joints.
Elevated blood inflammatory markers are present – ESR and CRP (erythrocyte sedimentation rate and C-reactive protein)23. Serum ‘rheumatoid factor’ may be conducted to exclude rheumatoid arthritis21. The genetic marker HLA-B27 has been found in over 50% of patients with psoriatic arthritis24.
Radiological imaging may show inflammatory joint changes (as opposed to erosive or degenerative joint destruction). Such radiological changes may not be notable in the early stages of AP. Enthesitis (inflammation of the tendon-bone junction) is characteristic of psoriatic arthritis and this symptom can aid in the differentiation of PA from other inflammatory joint conditions. Common enthesopathic sites include:
- The plantar fascia (pain on the sole of the heel bone)
- The finger flexor tendons (pain in the palm of the hand)
- The achilles tendon (the insertion of the achilles tendon onto the heel bone) 21.
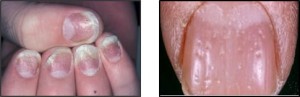 Psoriatic arthritis may also present with changes to the fingernails or toenails – pitting or ‘lifting’ may be pathognomonic25.
Psoriatic arthritis may also present with changes to the fingernails or toenails – pitting or ‘lifting’ may be pathognomonic25.
Treatment
In mild cases of psoriatic arthritis individuals may only be treated whilst the joints are painful and cease intervention when symptoms abate. In such cases NSAIDs such as ibuprofen or naproxen are commonly used.
Should desired outcomes not be managed with the first line of treatment DMARD medication is then trialled. Some cases require the combination of two different drugs.
Anti-tumor necrosis factor (anti-TNF) drugs have become available more recently and can treat both the arthritic and dermatological symptoms26.
Corticosteroid injections may be beneficial is mild to moderate cases though if joint degeneration is severe joint replacement surgery may be required.
Once symptoms of PA are controlled, exercise is recommended to maintain joint range of motion as well as strength and endurance of the surrounding musculature. Experienced physiotherapists should formulate targeted exercise rehabilitation programs.
Mr Joshua Clark (Kingsley Physiotherapy)
References
- http://medical-dictionary.thefreedictionary.com/arthropathy
- Cassidy, J. T., Levinson, J. E., Bass, J. C., Baum, J., Brewer, E. J., Fink, C. W., Hanson, V., Jacobs, J. C., Masi, A. T., Schaller, J. G., Fries, J. F., McShane, D. & Young, D. (1986). A study of classification criteria for a diagnosis of juvenile rheumatoid arthritis. Arthritis & Rheumatism, 29(2), 274-281.
- http://emedicine.medscape.com/article/970365-overview#a4
- http://emedicine.medscape.com/article/970365-overview#a5
- Kocher MS, Zurakowski D, Kasser JR. Differentiating between septic arthritis and transient synovitis of the hip in children: an evidence-based clinical prediction algorithm. J Bone Joint Surg Am. 1999 Dec. 81(12):1662-70.
- http://emedicine.medscape.com/article/970365-treatment
- Berek, C., & Kim, H. J. (1997, August). B-cell activation and development within chronically inflamed synovium in rheumatoid and reactive arthritis. In Seminars in immunology (Vol. 9, No. 4, pp. 261-268). Academic Press.
- Keat, A., Dixey, J., Sonnex, C., Thomas, B., Osborn, M., & Taylor-Robinson, D. (1987). Chlamydia trachomatis and reactive arthritis: the missing link. The Lancet, 329(8524), 72-74.
- Townes, J.M., Deodhar, A.A., Laine, E.S., Smith, K., Krug, H.E., Barkhuizen, A., Thompson, M.E., Cieslak, P.R. and Sobel, J. (2008). Reactive arthritis following culture-confirmed infections with bacterial enteric pathogens in Minnesota and Oregon: a population-based study. Annals of the Rheumatic Diseases, 67(12), pp.1689-1696.
- Tanaka, T., Kuwahara, Y., Shima, Y., Hirano, T., Kawai, M., Ogawa, M., Arimitsu, J., Hagihara, K., Narazaki, M., Ogata, A. and Kawase, I. (2009). Successful treatment of reactive arthritis with a humanized anti–interleukin‐6 receptor antibody, tocilizumab. Arthritis Care & Research, 61(12), pp.1762-1764.
- Kataria RK, Brent LH. Spondyloarthropathies. Am Fam Physician. 2004 Jun 15. 69(12):2853-60.
- Aho, K., Ahvonen, P., Lassus, A., Sievers, K., & Tiilikainen, A. (1974). HL‐A 27 in reactive arthritis. A study of yersinia arthritis and Reiter’s disease. Arthritis & Rheumatism, 17(5), 521-526.
- Savolainen E, Kettunen A, Närvänen A, Kautiainen H, Kärkkäinen U, Luosujärvi R, et al. (2009). Prevalence of antibodies against Chlamydia trachomatis and incidence of C. trachomatis-induced reactive arthritis in an early arthritis series in Finland in 2000. Scand J Rheumatol. 38(5):353-6.
- Kvien, T. K., Glennås, A., Melby, K., Granfors, K., Andrup, O., Karstensen, B., & Thoen, J. E. (1994). Reactive arthritis: incidence, triggering agents and clinical presentation. The Journal of Rheumatology, 21(1), 115-122.
- Wechalekar, M. D., Rischmueller, M., Whittle, S., Burnet, S., & Hill, C. L. (2010). Prolonged remission of chronic reactive arthritis treated with three infusions of infliximab. JCR: Journal of Clinical Rheumatology, 16(2), 79-80.
- Valtonen, V.V., Leirisalo, M., Pentikäinen, P.J., Räsänen, T., Seppälä, I., Larinkari, U., Ranki, M., Koskimies, S., Malkamäki, M. and Mäkelä, P.H. (1985). Triggering infections in reactive arthritis. Annals of the rheumatic diseases, 44(6), pp.399-405.
- Sieper, J., Rudwaleit, M., Braun, J., & van der Heijde, D. (2002). Diagnosing reactive arthritis: role of clinical setting in the value of serologic and microbiologic assays. Arthritis & Rheumatism, 46(2), 319-327.
- Valdimarsson, H., Baker, B. S., Jónsdóttir, I., Powles, A., & Fry, L. (1995). Psoriasis: a T-cell-mediated autoimmune disease induced by streptococcal superantigens?. Immunology today, 16(3), 145-149.
- Gladman, D. D., Shuckett, R., Russell, M. L., Thorne, J. C., & Schachter, R. K. (1987). Psoriatic arthritis (PSA)-an analysis of 220 patients. QJM, 62(2), 127-141.
- Taylor, W., Gladman, D., Helliwell, P., Marchesoni, A., Mease, P., & Mielants, H. (2006). Classification criteria for psoriatic arthritis: development of new criteria from a large international study. Arthritis & Rheumatism, 54(8), 2665-2673.
- Wilson, F. C., Icen, M., Crowson, C. S., McEvoy, M. T., Gabriel, S. E., & Kremers, H. M. (2009). Incidence and clinical predictors of psoriatic arthritis in patients with psoriasis: A population‐based study. Arthritis Care & Research, 61(2), 233-239.
- McGonagle, D., Gibbon, W., & Emery, P. (1998). Classification of inflammatory arthritis by enthesitis. The Lancet, 352(9134), 1137-1140.
- Graudal, N., Tarp, U., Jurik, A. G., Galløe, A. M., Garred, P., Milman, N., & Graudal, H. K. (2000). Inflammatory patterns in rheumatoid arthritis estimated by the number of swollen and tender joints, the erythrocyte sedimentation rate, and hemoglobin: longterm course and association to radiographic progression. The Journal of rheumatology, 27(1), 47-57.
- Sheehan, N. J. (2004). The ramifications of HLA-B27. Journal of the Royal Society of Medicine, 97(1), 10–14.
- Gladman D. D., Anhorn K. A., Schachter R. K., Mervart H. (1986). HLA antigens in psoriatic arthritis. J Rheumatol, 13(3), 586–592.
- Ogilvie, A. L. J., Antoni, C., Dechant, C., Manger, B., Kalden, J. R., Schuler, G., & Lüftl, M. (2001). Treatment of psoriatic arthritis with antitumour necrosis factor‐α antibody clears skin lesions of psoriasis resistant to treatment with methotrexate. British Journal of Dermatology, 144(3), 587-589.